While this and other vaccines have met the FDAs rigorous scientific standards for emergency use authorization as the first FDA-approved COVID-19 vaccine. 104 rows The FDA authorized the use under the emergency use authorization EUA for the Janssen COVID-19 vaccine of an additional batch of vaccine drug substance manufactured at the Emergent facility.

Covid Vaccine Moderna Applies For Full Fda Approval
The FDA determined that the available data for each vaccine provides clear.
Fda approved covid vaccine list. SAGE and EUL are complementary but independent processes. The Moderna COVID-19 Vaccine is a vaccine and may prevent you from getting COVID-19. The public is very confident that this vaccine meets the high standards of safety.
As of February 27 2021 large-scale Phase 3 clinical trials are in progress or being planned for two COVID-19 vaccines in the United States. Meets strict scientific standards for. An FDA slideshow presentation regarding Covid vaccines last year accidentally displayed a long list of possible adverse reactions to the vaccine including myocarditis seizures and even death.
Today the US. This has been known as the Pfizer-BioNTech Covid-19 vaccine and will now be marketed as Comirnaty for the prevention of Covid-19 disease. This is the first Covid-19 vaccine to get full approval from the FDA.
This vaccine and other vaccines are the first FDA-approved COVID-19 vaccines to be approved for emergency use by the FDA. 23 rows Adenovirus vaccine vector ChAdOx1-S recombinant Vaccines for human. SAGE has issued recommendations on Pfizer 8 January 2021 Moderna 25 January 2021 AstraZeneca 21 April 2021 and Janssen COVID 17 March 2021 vaccines as well as issued a framework for access and population prioritization roadmap.
While this and other vaccines have met the FDAs rigorous scientific standards for emergency use authorization as the first FDA-approved COVID-19 vaccine the public can be very confident that this vaccine meets the high standards for safety effectiveness and manufacturing quality the FDA requires of an approved product said Acting FDA Commissioner Janet Woodcock MD. The FDA thoroughly evaluated and analyzed the safety and effectiveness data for all of the authorized COVID-19 vaccines. While this and other vaccines have met the FDAs rigorous scientific standards for emergency use authorization as the first FDA-approved COVID-19 vaccine the public can be very confident that this vaccine meets the high standards for safety effectiveness and manufacturing quality the FDA requires of an approved product said Acting FDA Commissioner Janet Woodcock in a.
The slide showing the FDAs draft list of possible adverse event outcomes appeared briefly during a public meeting by the US Food and Drug Administrations Product Advisory Committee on Oct. Published by GulteDesk August 23 2021 The FDA granted approval to Pfizers Covid-19 vaccine for individuals 16 years and older. 9 rows COVID-19 Vaccine Moderna mRNA-1273 SARS-CoV-2 suspension for.
While this and other vaccines have met the FDAs rigorous scientific standards for emergency use authorization as the first FDA-approved COVID-19 vaccine the public can be very confident that this vaccine meets the high standards for safety effectiveness and manufacturing. FDA approval of this vaccine is a milestone in continuing the fight against the COVID-19 pandemic. Pentagon Announces Vaccine Mandate Theres a lot to keep track of.
Food and Drug Administration FDA approved vaccine to prevent COVID-19. Vaccines in Phase 3 Clinical Trials. FDA Grants First Full Approval for COVID-19 Vaccine.
There is no US. Novavax COVID-19 vaccine. In an announcement Monday the FDA said that the Pfizer vaccine first made available under EUA last December was the first COVID-19 vaccine to get full approval for people 16 years and over and will be marketed as Comirnaty.
List of COVID-19 Vaccines Authorized by the FDA. Food and Drug Administration amended the emergency use authorizations EUAs for both the Pfizer-BioNTech COVID-19 Vaccine and the Moderna COVID-19. The FDAs approval of this vaccine is a milestone as we continue to battle the COVID-19 pandemic.
Pfizer-BioNTech COVID-19 Vaccine BNT162b2 ChAdOx1-S recombinant COVID-19 Vaccine AstraZeneca SARS-CoV-2 Vaccine Vero Cell Inactivated Coronavac Sputnik V Gam-COVID-Vac COVID-19 Vaccine.
Every Vaccine And Treatment In Development For Covid 19 So Far

Health Experts Welcome Full Approval Of Pfizer Covid 19 Vaccine In Coming Weeks The Boston Globe
Fda Expected To Approve Third Covid Vaccine Dose For Immunocompromised People
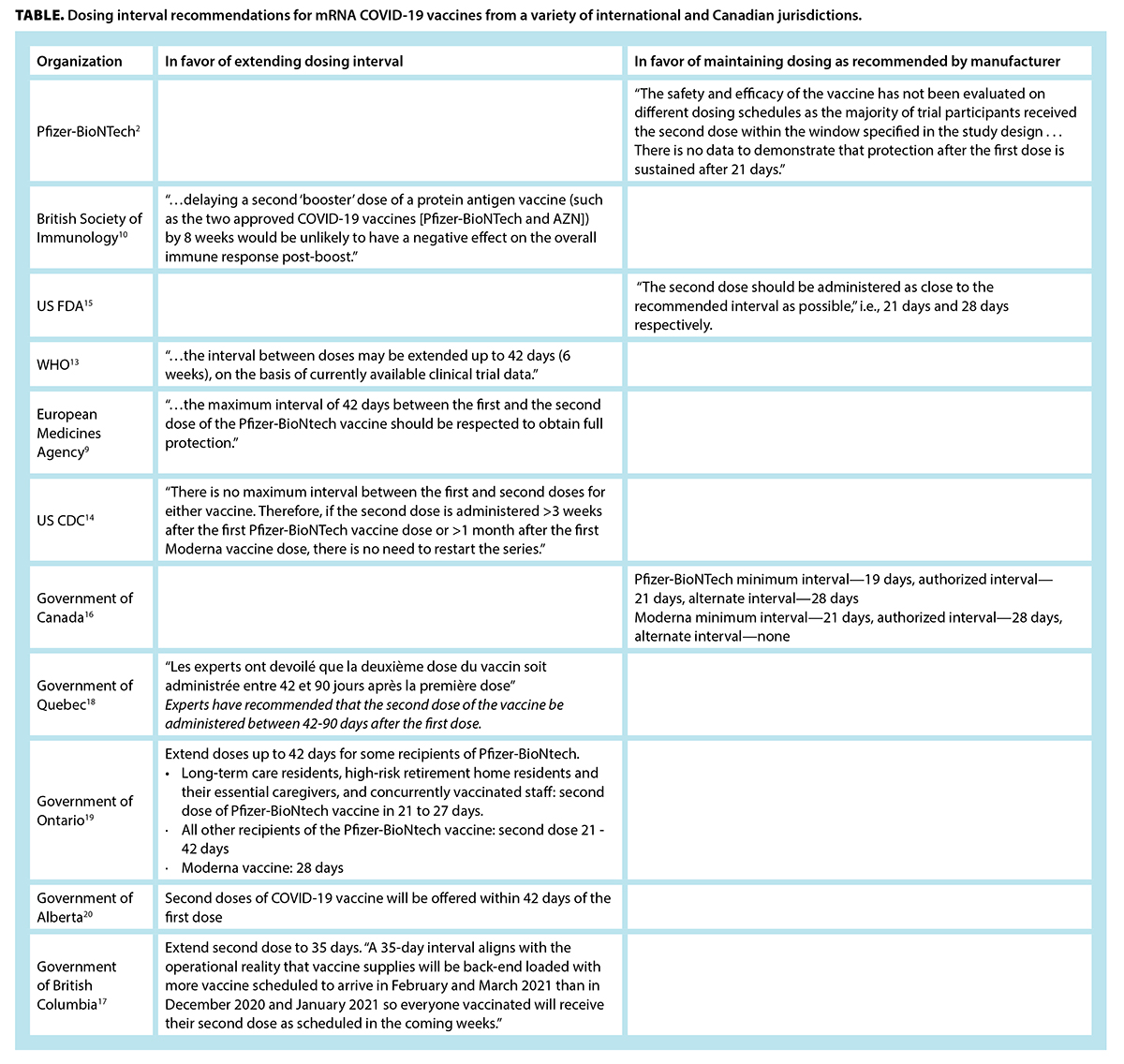
What Is The Evidence For Extending The Sars Cov 2 Covid 19 Vaccine Dosing Schedule British Columbia Medical Journal

Covid 19 Vaccinations What S The Progress Science In Depth Reporting On Science And Technology Dw 14 08 2021

Vaccine Communications Covid 19 Risk Communications Hub

Pfizer Inc On Twitter Breaking Today With Biontech Group We Submitted A Request To Us Fda To Expand Emergency Use Of Our Covid 19 Vaccine To Adolescents 12 To 15 Years Of Age Https T Co Quiqwq1kuz
Vaccine Resources Park County Co

Fda Advisory Panel Endorses Pfizer Biontech Covid 19 Vaccine

Fda Philippines Grants Emergency Use Authorization To Pfizer Biontech Covid 19 Vaccine Food And Drug Administration
Covid Vaccines What Full Fda Approval Means For You

Know Your Vaccines Vaccine Matrix Current Evidence Department Of Health Website

Know Your Vaccines Vaccine Matrix Current Evidence Department Of Health Website

Covid 19 Vaccine Need To Know Fliers Posters And Graphics Mass Gov

Lack Of Fda Covid Vaccine Approval Doesn T Matter Office For Science And Society Mcgill University